Definitions
Part 2
Energy
Energy is the capacity of a system to do work where "system" refers to any physical system, not just a refrigeration system.
Enthalpy
Enthalpy is the total amount of heat in one Lb. of a substance. It's units are therefore BTU/Lb. The metric counter part is kJ/kg. (kilo joules/kilogram)
Entropy
Entropy measures the energy dispersion in a system divided by temperature. This ratio represents the tendency of energy to spread out, to diffuse, to become less concentrated in one physical location or one energetic state. That spreading out is often done by molecules because molecules above absolute zero always have energy inside of them. That's why they are incessantly speeding through space and hitting each other and rotating and vibrating in a gas or liquid. Entropy is measured in BTU per lb. per °F.

Mollier Charts
Mollier charts are used in designing and analyzing performance of vapour compression refrigeration systems. Each refrigerant has it's own chart which is a graph of the Enthalpy of a refrigerant during various pressures and physical states. Mollier charts are also called Pressure-Enthalpy diagrams. Pressure is shown on the verticle axis, enthalpy is on the horizontal axis. You can compare Imperial versus SI Unit Mollier Charts by clicking on the buttons below the chart.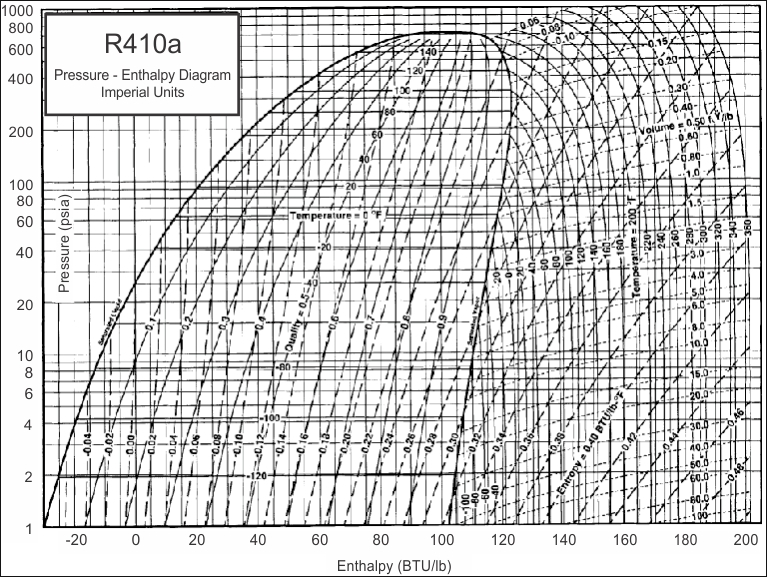
The above chart is for refrigerant MP39. You can see that pressure and enthalpy are the units on the verticle and horizontal axis.
Several other parameters are also shown on the chart including temperature, volume, saturation quality and entropy.
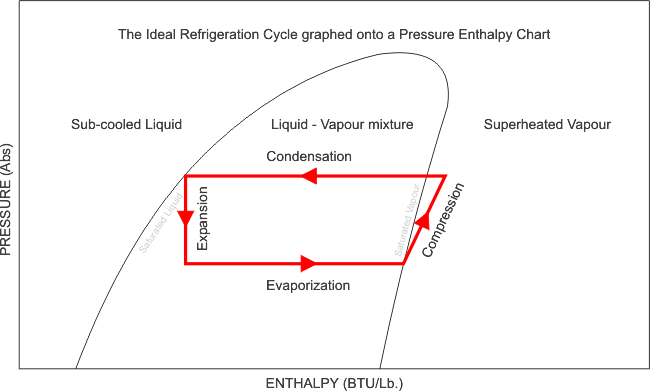
The series of graphics above shows how the refrigeration cycle is graphed onto the pressure-enthalpy chart and goes into details about how certain parameters can be determined from the chart.

